Today, researchers and physicians from the Mount Sinai Health System and its associated Icahn School of Medicine released a preprint* titled “SARS-CoV-2 infection induces robust, neutralizing antibody responses that are stable for at least three months.” The title of the paper suggests that there is antibody-mediated immunity to COVID-19 lasting at least three months after the onset of infection. Seen in the light of a a preprint released by Kings College London on July 11, however, this conclusion seems doubtful. Instead, it appears that the antibodies remain at high levels in most people for three months, but start losing their effectiveness in as little as two months.
The Mount Sinai Health System is in New York City. They used their own in-house antibody test to screen confirmed or suspected COVID-19 cases for antibodies that could be donated to treat critically ill patients in what is known as “convalescent plasma” treatments. They also allowed all employees to get tested if they chose to. Overall they screened 51,829 individuals, and 19,763 of them tested positive. These were overwhelmingly mild or moderate cases, as only about 5% required hospitalization or emergency room evaluation.
They acknowledged that they could be missing mild to moderate cases that did not develop antibodies, but they cited other studies showing that 99% of 568 PCR-confirmed cases developed antibodies and 95% of 2,347 patients who self-reported being PCR-positive developed antibodies. They concluded from this that they weren't missing very large portions of the mild-to-moderate cases.
As a side note, if we put this in context of the three immunotypes we covered yesterday, it would seem the vast majority of mild to moderate cases are immunotype 2, who have very well-trained B cells and a preponderance of memory cells that provide a lasting source of antibodies. While the Science paper covering the three immunotypes associated immunotype 3, which fails to launch either a T cell or antibody response, with less severe disease within hospitalized patients, and while this immunotype constituted 20% of the hospitalized patients, it does not seem to be prevalent among cases so mild as to never enter the hospital.
While it is too early to say for sure, it seems like immunotype 2 is the ideal and by far and away the most common immunotype; immunotype 1 is vulnerable to the cytokine storm and severe complications; immunotype 3 is perhaps in some limbo that gets sick very slowly and stays sick for a very long time.
While the Mount Sinai paper included 19,763 cases, only 121 of them were tested at two time points, day 30 after symptom onset and day 82 after symptom onset. In this subset, overall antibody titers remained nearly identical between day 30 and day 82.
In those with very high titers, they tended to decline somewhat. In those with very low titers, they tended to increase somewhat. They suggest this is because those with mild cases take longer to maximize their antibody response. While it could represent something physiological like that, it could also easily reflect the statistical artifact known as regression to the mean.
Now, the title of the paper says these are “robust, neutralizing antibody responses that are stable for at least three months.” I suppose they derive “three months” from 82 days plus some incubation period. But how do they know the neutralization lasts three months? They don't.
They claim it as follows. 120 samples, presumably all from the first timepoint though this is not clearly stated, were tested for their ability to neutralize SARS-CoV-2 replication in isolated cells. The concentration of antibodies correlated with the ability to neutralize the virus with a value of r=0.87. The square of this value tells you the percentage of variation in one variable explained by the other. This means that the concentration of antibodies could explain 76% of their ability to neutralize the virus.
From this, they then assume that this correlation persists across time points and assume that because antibody titers remained stable from day 30 to day 82 that neutralization ability also remained stable.
But did it?
If we consider this paper in the light of the Kings College paper, probably not.
The Kings College paper used sequential samples from 65 individuals with PCR-confirmed SARS-CoV-2 infection and 31 healthcare workers with positive antibodies up to 94 days after the onset of symptoms.
Like Mount Sinai, Kings College used their own in-house antibody assay. Somewhat differently, they tested the ability of the serum samples to neutralize the virus using a synthetic virus based on HIV that incorporates the spike protein into its membrane. The spike protein is the protein on the viral surface that it uses to bind to and infect cells. Since the neutralizing antibodies are all to the spike protein or to part of the spike protein, this assay should accurately reflect neutralization potential and is somewhat safer to perform since it does not involve handling live SARS-CoV-2 virus.
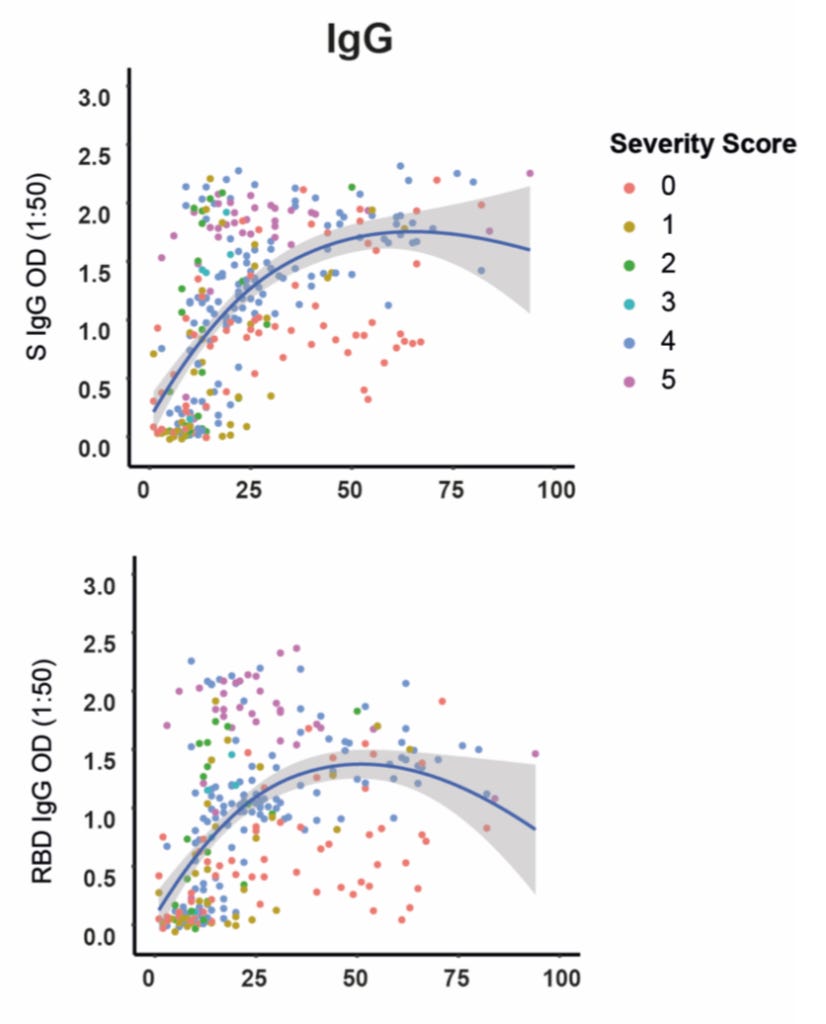
The most important antibodies for long-term immunity are IgG antibodies to the spike protein and its receptor-binding domain (RBD), which is the portion of the spike protein that binds to ACE2 on the human cell surface. As you can see below, there was some hint of a decline by day 94, but the antibodies remained at very high levels:
Antibodies to the spike protein are on top and antibodies to the RBD are on the bottom. The colored dots represent the severity level shown in the upper right. The blue line represents the average. The gray shaded area around the line represents the 95% confidence interval. When the gray area hugs the line tightly, it is because there is a 95% probability that the true value lies close to the line. When the gray area disperses widely, it is because there is less data and the 95% probability is that the true value lies possibly further from the line, anywhere within the shaded area.
IgG to the spike protein barely decreased by day 94, and the 95% confidence interval even includes a continued increase. IgG to the spike protein's RBD decreased somewhat more, but the 95% confidence interval still includes it remaining nearly the same.
Contrast this with the virus-neutralizing effect of the serum:
The mean falls almost back to the level at day 0. The 95% confidence interval excludes anything other than a large decline in neutralization.
The Bottom Line
While antibodies seem to persist at high levels over three months, they appear to begin losing effectiveness, on average, by day 50, and to lose most of their effectiveness by the three-month point.
While more studies with larger samples, longer followup, and comparisons between different antibody assays and virus-neutralization assays will be needed to come to firm conclusions, we should tentatively conclude as follows:
High antibody levels suggest a high probability of immunity in the short-term, but they become much less reliable indicators of immunity after two months.
As we covered two days ago, there may be a long-lasting T cell response that provides immunity even when the effectiveness of the antibodies wanes. We cannot assume that low neutralization at 94 days means low immunity at 94 days. However, we also cannot assume the opposite. These results are bearish for the possibility of herd immunity and vaccine effectiveness. It remains within the realm of possibility that COVID-19 could recur as easily as common cold viruses do, and we desperately need to study exactly what constitutes immunity and what can be used as reliable markers of immunity.
This is not a message of despair. Good nutrition and proper precautions to avoid the virus go a long way towards protecting us. This is instead a message that it is not yet time to stop taking this seriously. In particular, long-lasting antibodies cannot be used to dismiss the need to continue taking COVID-19 seriously. The hope lies in continuing to learn how to protect ourselves and each other, building constantly on what we have already learned.
Stay safe and healthy,
Chris
Please Support This Service
These research updates are made possible by purchases of The Food and Supplement Guide to the Coronavirus. The guide contains my most up-to-date conclusions about what we should be doing for nutritional and herbal support on top of hygiene and social distancing for added protection. Due to the absence of randomized controlled trials testing nutritional or herbal prevention, these are my best guesses for what is likely to work without significant risk of harm, based on the existing science. By purchasing the guide, you are enabling me to continue devoting my skills to the most important issue we now face. I am genuinely grateful for your contribution. You can purchase the guide using this link.
You can get the guide for free if you pre-order my upcoming book, Vitamins and Minerals 101: How to Get the Nutrients You Need on Any Diet (to be released when the COVID-19 crisis subsides) or when you join my membership program, the CMJ Masterpass, which is meant to help people with significant health and wellness expenditures consistently save money by returning marketing cost of the companies involved back to the members as rebates.
For other ways to support my work, please see here.
Disclaimer
I am not a medical doctor and this is not medical advice. I have a PhD in Nutritional Sciences and my expertise is in conducting and interpreting research related to my field. Please consult your physician before doing anything for prevention or treatment of COVID-19, and please seek the help of a physician immediately if you believe you may have COVID-19.
Subscribe
If you aren't subscribed to the research updates, you can sign up here.
Archive
You can access an archive of these updates here.
Comments and Questions
To leave a comment or question, please use the Facebook post for this newsletter.
*Footnotes
* The term “preprint” is often used in these updates. Preprints are studies destined for peer-reviewed journals that have yet to be peer-reviewed. Because COVID-19 is such a rapidly evolving disease and peer-review takes so long, most of the information circulating about the disease comes from preprints.